Challenges Faced Without Change Management System
Every progress is a change. Getting rid of old processes, or obsolete systems to bring new one is a change. Organisations implement change to improve processes, enhance their productivity, and increase customer satisfaction.
ISO 9001 identifies change as a response to customer complaints or product failures to identify opportunities for improvement or innovation.
But sometimes, this change is chaotic, mostly if done through paper/spreadsheets based methods. What organisations need is a Change Management Software to bring this change smoothly.
QualityPro is one such software having amazing change management capabilities. It handles change from start to the end in a congenial manner. Apart from recording and managing the change, the software helps to ensure that the changes are reviewed, verified, validated, and approved, thereby meeting the compliance. With QualityPro, change is no more a nightmare. It’s a spree of betterment.

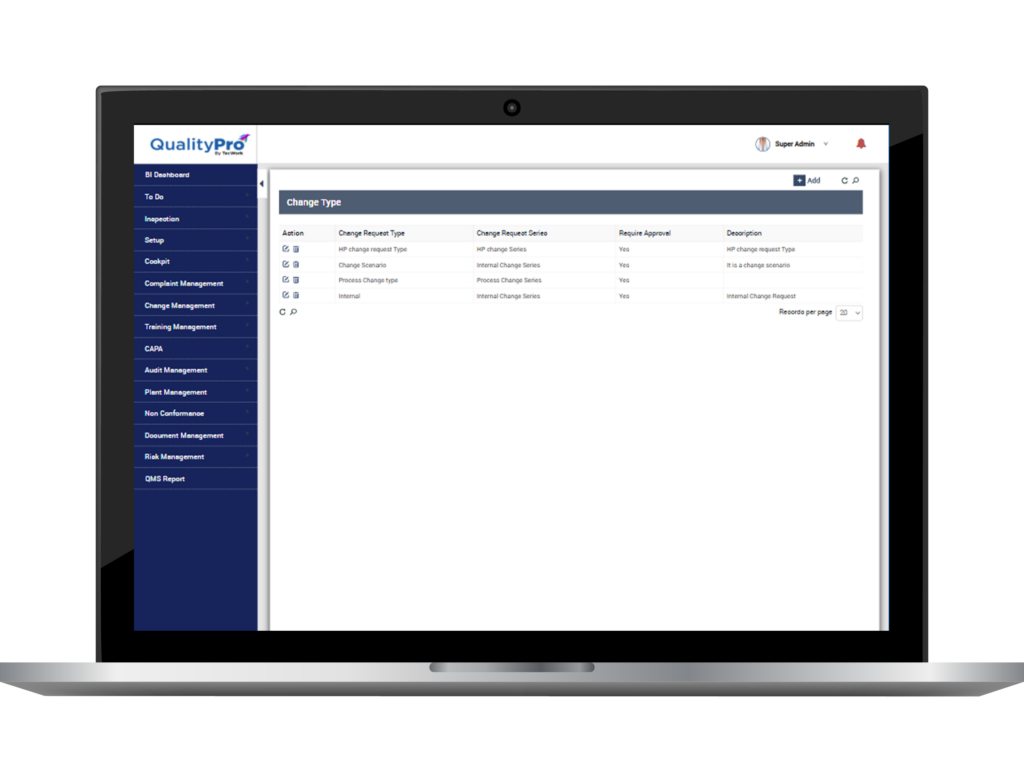
Centralised Control Over Change
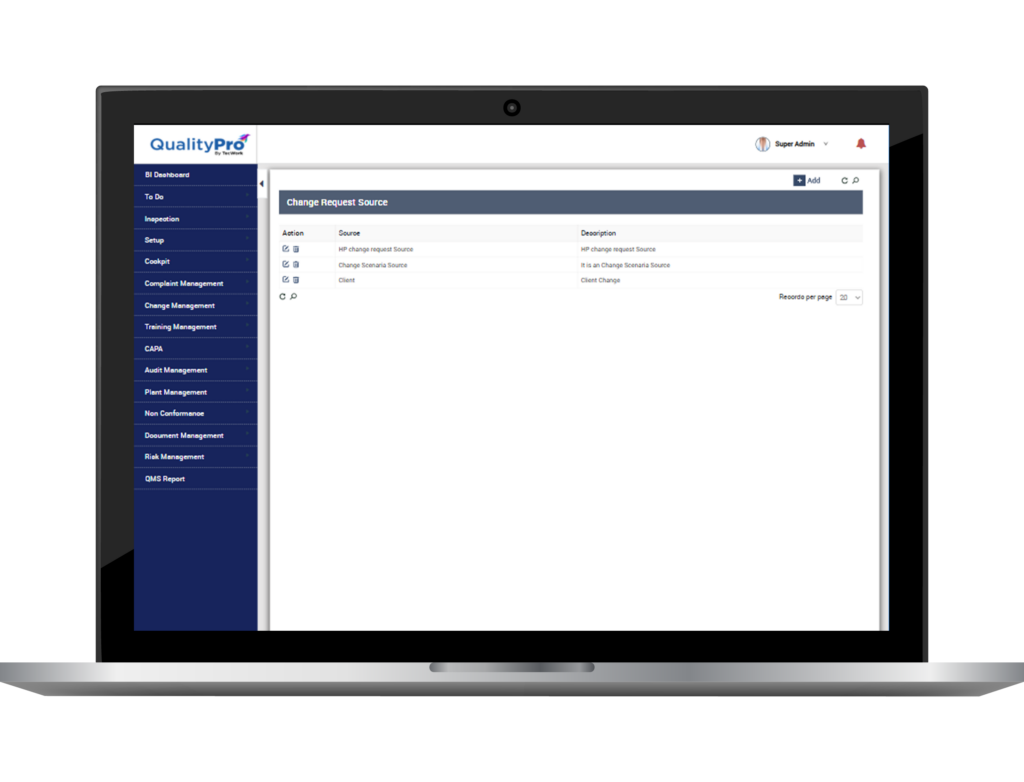
- Raise change request, define its source, its type/category, document it and save it centrally. Define reason of change and describe change.
- Define plan of implementation and track it coming into action.
- Identify departments affected by the change and identify document type to be changed.
- Replace error prone paper-based system with automated error-free digitised system.
- Evaluate proposed change by measuring the cost associated with the change.
- Identify the employees who will require training with the change and schedule training for those employees.
Your path to better quality management starts here
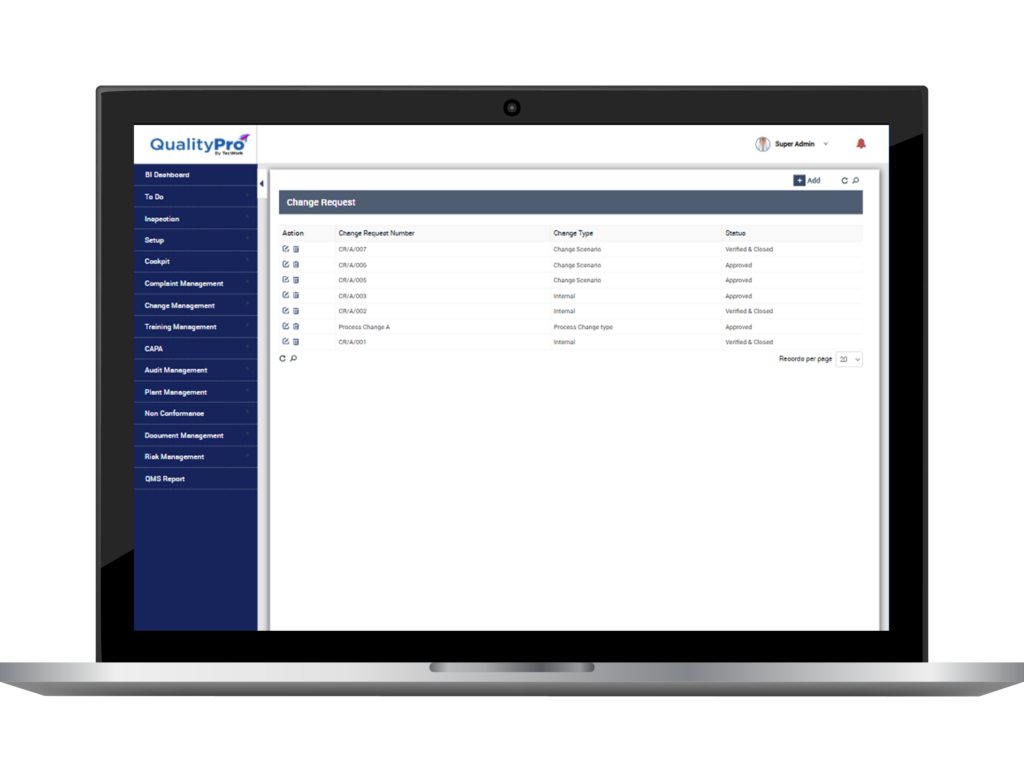
Bring Accountability and Traceability to Life
- Track who, when, how, what changes were made in the document.
- Define procedural workflows with authorised access.
- Comprehensively manage change requests by outlining the purpose of the change, identifying its source, detailing the execution plan, and documenting closure. Additionally, efficiently track change versions for enhanced control
- Retrieve data related to change, like number of change versions, training personnel, source of change etc., and share the data with concerned stakeholder.
Don’t just take our word for it—Experience QualityPro firsthand
Bring Accountability and Traceability to Life
- Track who, when, how, what changes were made in the document.
- Define procedural workflows with authorized access.
- Comprehensively manage change requests by outlining the purpose of the change, identifying its source, detailing the execution plan, and documenting closure. Additionally, efficiently track change versions for enhanced control
- Retrieve data related to change, like number of change versions, training personnel, source of change etc and share the data with concerned stakeholder.

Ensure Compliance
- Manage and store all documents systematically, keep them easily accessible, and track all the know-how of the change.
- Keep record of all the actions taken, make sure all steps are followed to ensure successful change implementation.
- Validate, evaluate and approve the impact of change before closing it.
- Store versions of document and purge old or obsolete documents to enforce only the latest and approved one.
Benefits of Change Management Module
- Automates all the tasks pertaining to change management.
- Offers easy integration between the change management process and training.
- Provides support to complete change management cycle.
- Provides complete visibility into change management process.